But who has time for all that?! These days, you can simply screen dozens of candidates by reading their dating profiles on matchmaking web sites or apps like Tinder. This modern form of mate selection is unique to humans; imagine if other creatures in the natural world, like parasites, had to write dating profiles…
Toxoplasma gondii
 |
Hanging with my BFFs in a tissue cyst.
We call ourselves "The Brady Bunch"!
Photo by David Ferguson (via EurekAlert)
|
Do you love cats? So do I! They’re my favorite animal,
although I can weasel my way into any vertebrate animal that I want to,
including weasels. That’s one of the reasons why I’m called “the most
successful parasite on Earth.” I’m the clever parasite that has learned to
manipulate the brains of rodents so that they become fearless morons around
felines. Normally, mice and rats scurry away from the scent of a cat, but not
when I’m in their head!
What turns me on? Long, romantic walks through the hollows
of a cat’s innards. I like to groove under the moist sheets of their intestinal
epithelium to the musical stylings of Cat Stevens. If we have kids, I promise
to be a good parent and read Calvin and
Hobbes to them all night long. I’ll be sure to kiss them goodbye before
sending them out into the world to contaminate litter boxes, sandboxes,
gardens, yards, and streams. Before long, our progeny will be inhaled or
ingested by unsuspecting animals.When I get into something that is not a cat, I get bored rather quickly and go to sleep. You can call me bradyzoite when I’m napping. Life in my intermediate host isn’t all that bad. I can pick pretty much whatever cell type I want and make it my room. The neurons in the brain are ideal because the pesky immune system tends to leave that organ alone, so I get plenty of peace and quiet. I just chill and wait for that animal to get eaten, hopefully by a cat so I can get my groove on again! What if another type of animal eats me instead? No biggie. I’m a patient parasite and will simply wait it out in another intermediate host.
Like I said before, if I landed in a rodent I know how to scramble their tiny brains to increase their chances of getting eaten by a hairball-coughing feline. The human brain is a tad more complex and taking me a little longer to figure out. While knocking around in a human head, I might have increased the risk of some people to develop schizophrenia or rage disorder. But ultimately, I’m trying to rewire the human brain so they leap into lion cages at the zoo.
In my spare time, I love to devour books instead of organ meat. My favorite books include Cat’s Cradle, The Pink Panther, The White Tiger, and of course The Cat in the Hat. I’m also writing my own book. It’s called If You Give A Mouse Toxoplasma…
Schistosoma mansoni
 |
Come swim with Schisto!
Photo: http://schaechter.asmblog.org/.a/
6a00d8341c5e1453ef014e875d2f3e970d-popup
|
Escargot, anyone? My name is Schistosoma, but you can call
me Schisto. I live in parts of South America and the Caribbean, Africa, and the
Middle East. I hope you don’t think I’m being too fresh, but I’d love to start
our date by skinny dipping in my favorite freshwater lake. After we’re done
frolicking in the water, we’ll sneak into some snails and develop into cercariae.
What? You’ve never been a cercariae before? Have no fear, my darling, I will
teach you how to become one. Once we’re cercariae, we’ll break out of the snail
and search for the definitive stop on our romantic adventure: an unsuspecting
human swimming in our waters.
The cool thing about becoming cercariae is that we’ll look
like a mermaid. We’ll gain a gorgeous forked tail that will help us swim around
and find a suitable human to invade. I like to hum the theme to Jaws as I make
my approach to the human creature! Do you know how many people are attacked by sharks each year? Only 75. I've infected well over 200 million people, but sharks get the scary theme song...go figure!I think you’ll be surprised how easy it is to burrow into a human's skin – I prefer to enter through a hair follicle. They don’t feel a thing. Once we get inside a human, we can ditch our tails and I’ll give you a grand tour. After a few days gallivanting through the skin, we’ll hang out in the lungs, go through the heart, and then enjoy a bloodmeal as we take a ride in the circulatory system to the liver. This is the stop I find most arousing, and I’ll ask you to pair-bond with me. If you accept, we’ll celebrate by making our way to the veins draining the colon.
Why the veins of the colon? I’m glad you asked, my pet! You see, the colon is where the human stores his waste until he can’t hold it in any longer. We can easily send our eggs into his colon, giving our kids a free ride back out into the water so they can find snails of their own one day. It’s a strategy not unlike the one used by Han Solo in The Empire Strikes Back when he evaded the Star Destroyer by making it appear his ship was just a part of the Imperial garbage.
I think you’ll find that the chemistry between us is no fluke, but rather truly meant to be.
Trypanosoma cruzi
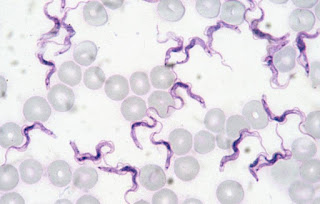 |
Come cruzi with me! I'm the cute wavy purple things!
Photo: Wikipedia Commons
|
Once under the human’s skin, we’ll transform from trypomastigotes into amastigotes while inside the host’s cells. I hope you don’t think I’m being too prudish, but I’m really not all that into sex. I’d prefer that we multiply on our own, but how about this…we can watch each other divide!
After we make clones of ourselves, there will be too many for the host cell to hold. I just love it when a host cell pops, don’t you? As trypomastigotes again, we’ll be free floating in the blood, where we will hitch a ride when the next hungry bug comes along to “kiss” our human host.
I just hope the kids we leave behind don’t cause trouble. Most of the time when I go through a human, my kids get all rowdy and start having a bunch of kids of their own. The extensive damage they leave in their wake can cause serious problems for the human host, which they call Chagas disease.
While waiting for a kissing bug to pick me up, I enjoy listening to music. Some of my favorite songs include Kiss Me Deadly, Love Bites, and Blow Me (One Last Kiss).
Plasmodium falciparum
 |
I'm a little camera shy, but I like these plushies of me
as they show my softer side!
Photo: Giant Microbes
|
If you have a fetish for vampires or other blood-sucking
creatures, I am the parasite for you! My name is Plasmodium, but most people know me as malaria, which means “bad
air.” I hasten to clarify: I do not suffer from flatulence or rancid breath.
Before people realized I was a parasite, they attributed the cause of malaria
to breathing in “bad air.”
Two of my favorite things in life are blood and sex. I use
humans for blood and Anopheles
mosquitoes for sex. You might not think that there is enough room in the gut of
a mosquito to have a lot of great sex, but give me a chance and I’ll show you
that size isn’t everything. After the love making, we’ll take a lovely stroll
up to the mosquito’s salivary glands and take a little nap before dinner. While
we’re in the salivary glands, you can call me sporozoite.The mosquito will be our limo to a fine human restaurant where the blood flows like wine. We will get our wake-up call when the mosquito bites a person; then hang tight while we take an exhilarating slide down her proboscis and into a red river. After a quick pit stop in the liver to transform into merozoites and put on our bibs, we’ll jump back into the red river and take our pick at which blood cell we’d like to dine at first. All the hemoglobin you can eat! We will be the envy of Count Dracula!
In humans, red blood cells carry oxygen around the body, so as we destroy them, our human host will soon feel woozy, suffering from anemia, chills, and fever. But have no fear, as I’ll send out an SOS that changes our victim’s scent to be more attractive to mosquitoes. Before you know it, we’ll be pulled up into a fresh mosquito for some more amore.
I’m also a huge movie buff. My favorite movies are The Mosquito Coast, There Will Be Blood, Jungle Fever, and Red River.
Contributed by: Bill Sullivan
Follow Bill on Twitter. 








